- Scarring from a prior heart attack or other causes: A heart that's scarred or enlarged from any cause is prone to develop life-threatening ventricular arrhythmias. The first six months after a heart attack is a particularly high-risk period for sudden cardiac arrest in patients with atherosclerotic heart disease.
- A thickened heart muscle (cardiomyopathy) from any cause (typically high blood pressure or valvular heart disease) — especially if you also have heart failure — can make you more prone to sudden cardiac arrest.
Read more about cardiomyopathy
Heart medications: Under certain conditions, various heart medications can set the stage for arrhythmias that cause sudden cardiac arrest. Paradoxically, antiarrhythmic drugs used to treat arrhythmias can sometimes produce lethal ventricular arrhythmias even at normally prescribed doses. This is called a "proarrhythmic" effect. Regardless of whether there's organic heart disease, significant changes in blood levels of potassium and magnesium (from using diuretics, for example) also can cause life-threatening arrhythmias and cardiac arrest. - Electrical abnormalities: Certain electrical abnormalities such as Wolff-Parkinson-White syndrome and long QT syndrome may cause sudden cardiac arrest in children and young people.
Read more about Wolff-Parkinson-White syndrome
Read more about long QT syndrome
Read special considerations about cardiac arrest in children - Blood vessel abnormalities: Less often, inborn blood vessel abnormalities, particularly in the coronary arteries and aorta, may be present in young sudden death victims. Adrenaline released during intense physical or athletic activity often acts as a trigger for sudden cardiac arrest when these abnormalities are present.
- Recreational drug use: In people without organic heart disease, recreational drug use is a cause of sudden cardiac arrest.
|
Sudden cardiac arrest may be caused by almost any known heart condition. Most cardiac arrests occur when the diseased heart's electrical system malfunctions, producing an abnormal rhythm such as ventricular tachycardia or fibrillation. Some cardiac arrests are caused by extreme slowing of the heart's rhythm. All these events are called life-threatening arrhythmias.
29 Comments
Cardiac arrest strikes immediately and without warning. Here are the signs:
What is cardiac arrest?
Cardiac arrest, also known as sudden cardiac arrest, is the abrupt loss of heart function in a person who may or may not have diagnosed heart disease. The time and mode of death are unexpected. It occurs instantly or shortly after symptoms appear. Each year, nearly 360,000 emergency medical services-assessed out-of-hospital cardiac arrests occur in the United States. Is a heart attack the same as cardiac arrest? No. The term "heart attack" is often mistakenly used to describe cardiac arrest. While a heart attack may cause cardiac arrest and sudden death, the terms don't mean the same thing. Heart attacks are caused by a blockage that stops blood flow to the heart. A heart attack (or myocardial infarction) refers to death of heart muscle tissue due to the loss of blood supply, not necessarily resulting in the death of the heart attack victim. Cardiac arrest is caused when the heart's electrical system malfunctions. In cardiac arrest death results when the heart suddenly stops working properly. This may be caused by abnormal, or irregular, heart rhythms (called arrhythmias). A common arrhythmia in cardiac arrest is ventricular fibrillation. This is when the heart's lower chambers suddenly start beating chaotically and don't pump blood. Death occurs within minutes after the heart stops. Cardiac arrest may be reversed if CPR (cardiopulmonary resuscitation) is performed and a defibrillator is used to shock the heart and restore a normal heart rhythm within a few minutes. Initial Assessment1-1. WHAT ARE "VITAL SIGNS"?
Following the initial assessment and control of any immediate life threats, you will begin a more thorough assessment of your patient. Two essential elements of this assessment will be measuring vital signs and obtaining a medical history. Vital signs are measurable life signs. The term "vital signs" usually refers to the patient's temperature, pulse, breathing, and blood pressure. Because they reflect the patient’s condition, you must take them early and repeat them often. The medical history includes information about the present medical problem and facts about the patient that existed before the patient required emergency medical response. This information can affect the treatment you give. It is called a SAMPLE history because the letters in the word SAMPLE stand for elements of the history. 1-2. INITIAL ASSESSMENT AND SAMPLE HISTORY a. A thorough, properly sequenced rapid assessment is essential to identifying a patient’s needs and providing proper emergency care. To assess a patient, the combat medic must gather, evaluate, and record key information including the patient’s vital signs, injuries, and symptoms and the conditions leading to the illness or injury. The combat medic must learn the history of what happened before and since the accident or medical problem occurred and learn the patient's past medical history and overall health status. Assessment is a process which must be taught in steps in order to establish good assessment habits and a systematic approach in order to avoid missing important injuries or illnesses. In the field, many aspects of the patient assessment may be done simultaneously. b. Assessment is one of the most complex skills that must be learned to be an effective combat medic. During the assessment, the combat medic must use his eyes, ears, nose, hands, and a few basic medical instruments to obtain essential information about the patient. 1-3. ESTABLISH PATIENT CONTACT a. After assessing the scene for safety and determining the need for additional help, the number of patients, the mechanism of injury or nature of illness, and considering the need for cervical spine immobilization, the combat medic must make contact with the patient. The patient is assessed for ABC’s and his level of consciousness; then you can begin questioning your patient for vital information about the current medical problem with which you are concerned. This is the "chief complaint." b. The chief complaint is the major signs, symptoms, or events that caused the illness or injury. Symptoms are conditions that the patient feels and tells you about, such as dizziness or particular pain. Symptoms are the subjective information you obtain from you patients. Signs can be seen, heard, felt, smelled, or measured, such as wounds, external bleeding, deformities, breathing rate, and pulse. You must be able to record and report how and when the signs and symptoms began. c. Initial assessment is a rapid evaluation of the patient’s general condition to identify any potentially life-threatening injuries or conditions. (1) Repeated vital signs will be compared to the baseline set. (2) Vital signs are key indicators used to evaluate and determine the patient’s overall condition. Because key indicators include quantitative (numeric) measurement, vital signs always include breathing, pulse, and the blood pressure. (a) The first vital sign is breathing. Breathing is discussed in Lesson 4.(b) The second vital sign is the pulse. The pulse is discussed in Lesson 3.(c) The third vital sign is the blood pressure. Blood presser is discussed in Lesson 5.(d) Other key indicators include:
The skin is an easily observed indicator of the peripheral circulation and perfusion, blood oxygen levels, and body temperature. The skin color, temperature, and condition are good indicators of the patient's condition and circulatory status. They may also be good initial indicators of heat or cold injuries. This initial indicator should always be confirmed, when time permits, with a core body temperature (see Lesson 2). a. Color. (1) Many blood vessels lie near the surface of the skin.(2) Pigmentation in individuals will not hide changes in the skin’s underlying color.(3) In lightly pigmented individuals, skin normally has a pink color.(4) In patients with deeply pigmented skin, changes in skin color may only be apparent in certain areas, such as the fingernail beds, the lips, the mucous membranes in the mouth, the underside of the arm and hand, and the conjunctiva of the eye.(5) Poor peripheral circulation will cause the skin to appear pale, white, ashen, gray, or waxy and translucent like a white candle. These skin colors can also appear in abnormally cold or frozen skin.(6) When the blood is not properly saturated with oxygen, it changes to a bluish color. Skin over the blood vessels appears blue or gray, a condition called cyanosis.(7) Red skin will result from carbon monoxide poisoning, significant fever, heatstroke, sunburn, mild thermal burns, or other conditions in which the body is unable to properly dissipate heat.(8) Color changes may also result from chronic illness. Liver disease dysfunction may cause jaundice, resulting in a yellow cast to the skin.b. Temperature. (1) Normally, the skin is warm to the touch.(2) The skin feels hot with significant fever, sunburn, or hyperthermia.(3) The skin feels cool in early shock, profuse sweating, heat exhaustion, and profound hypothermia and/or frostbite.(4) Feel the patient’s forehead with the back of your ungloved hand to determine marked fever.c. Moisture. (1) The skin is normally dry.(2) Wet, moist, or excessively dry and hot skin is considered abnormal.(3) In descriptions of the skin, it is usually listed as color, condition, and temperature (CCT).d. Capillary Refill. Capillary refill can be assessed as part of the evaluation of the skin. (1) Capillary refill is used to evaluate the ability of the circulatory system to restore blood to the capillary system (perfusion). Capillary refill is used primarily in the assessment of pediatric patients. Refill time in adults is not considered as accurate due to differences in circulation from medications and various other factors. This can still be used as a simple test of perfusion to the extremities, but many factors must be considered, such as the age of the patient and the environment (cold will decrease capillary refill time). (2) Capillary refill is evaluated at the nail bed in a finger. (a) Place your thumb on the patient’s fingernail and gently compress.(b) Pressure forces blood from the capillaries.(c) Release the pressure and observe the fingernail.(d) As the capillaries refill, the nail bed returns to its normal deep pink color.(e) Capillary refill should be both prompt and pink.(f) Color in the nail bed should be restored within 2 seconds, about the time it takes to say "capillary refill."1-5. ASSESSING LEVEL OF CONSCIOUSNESS Level of consciousness should also be assessed upon initial contact with your patient and continuously monitored for changes throughout your contact with the patient. a. AVPU. The AVPU scale is a rapid method of assessing LOC. The patient's LOC is reported as A, V, P, or U. (1) A: Alert and oriented. (a) Signifies orientation to person, place, time, and event. Ask your patient simple open ended questions that can not be answered with yes or no to determine the LOC. For example, "Where are you right now?" and "What time is it?" Do not ask your patient, "Do you know were you are right now?" since this can be answered with a yes or no. (b) If the patient is alert, you can reported your results as a patient oriented score from 1 (lowest) to 4 (highest), noting any areas not oriented to. For example, you can state the patient is "A and O X 4" (fully alert and oriented) or "A and O x 2 and does not know time and place." (2) R: Responds to verbal stimulus. This indicates that your patient only responds when verbally prompted. It is also important to note if the patient makes appropriate or inappropriate responses. If you ask your patient, "What is your name?" and he responds with, "Flaming monkeys," this would be an inappropriate response and shows that although he responds to verbal, he is not appropriately oriented. (a) Response to normal voice stimuli.(b) Response to loud voice stimuli.(3) P: Responds to pain. (a) Use if patient does not respond to verbal stimuli.(b) Gently but firmly pinch patient’s skin.(c) Note if patient moans or withdraws from the stimulus.(4) U: Unresponsive. (a) If the patient does not respond to painful stimulus on one side, try the other side.(b) A patient who remains flaccid without moving or making a sound is unresponsive.NOTE: Anything below Alert is unconscious; from there we need to determine how unconscious the patient is. A patient can be unconscious with response to stimuli or unresponsive. b. Glasgow Coma Scale. The Glasgow Coma Scale is an assessment based on numeric scoring of a patient’s responses based on the patient's best response to eye opening, verbal response, and motor response. The patient's score (3 to 15) is determined by adding his highest eye opening, verbal response, and motor response scores. (1) Eye opening (1 to 4 points). (a) Spontaneous: E4. Eyes are open and focused; the patient can recognize you and follow eye movements.(b) To voice: E3. The patient opens his eyes when spoken to or when directed to do so.(c) To pain: E2. The patient opens his eyes when given some sort of painful stimuli.(d) None: E1.(2) Verbal response (1 to 5 points). (a) Oriented: V5. The patient can talk and answer questions about his location, time, and who he is. In some situations, it is also appropriate to question the patient to see if he is oriented to the event that led him to be in his current condition.(b) Confused: V4. The patient can talk and speak coherently, but is not entirely oriented to person, place, time, and event.(c) Inappropriate words: V3. The patient answers with some sort of inappropriate response to the question that was asked or answers with excessive use of profanity that is not associated with anger toward the event.(d) Incomprehensible words: V2. Unintelligible words or sounds.(e) None: V1(3) Motor response (1 to 6 points). (a) Obeys command: M6. The patient can follow appropriate commands or requests. It is also important to asses the patient for the ability to follow commands across the central plane of the body. For example, the command, "Please touch your left shoulder with your right arm," helps to ensure the patient can cross the hemispheres of the brain since the left and right sides of the body are controlled by the opposite sides of the brain.(b) Localizes pain: M5. Can the patient localize the pain that he is feeling? If you elicit a pain response by pinching of squeezing the right side, watch for the patient to reach across with the opposite arms to check for cross body localization.(c) Withdraws to pain: M4. This indicates a correct pain response. The body should withdraw away from the pain and not towards it.(d) Flexion (decorticate posturing): M3. This is an abnormal posturing usually caused by severe brain trauma. The body curls into a protective posture by flexing the arms into the chest.(e) Extension (decerebate posturing): M2. In this form of posturing, the body is abnormally extended. The arms and legs may be extended and very rigid or difficult to move.(f) None: M1.c. PEARRL. Use the guide PEARRL when assessing the pupillary response of the patient's eyes. (1) P: Pupils. Are they both present? What is their general condition? (2) E: Equal. Are both pupils the same size? Unequal pupils can indicate a head injury causing pressure on the optic nerve. There is a small percent of the population that has unequal pupils normally, so a good patient history is critical. (3) A: And. (4) R: Round. (5) R: Regular in size. (6) L: React to light. Both eyes should be assessed twice for reaction to light. The first time the light is shined in the right eye, for example, you should watch the right eye for reaction, the second time the left eye should be watched to ensure sympathetic eye movement is present. (both eyes are doing the same thing at the same time). d Vital Signs. (1) The first set of vital signs establishes an important initial measurement of the patient’s condition and serves as a key baseline.(2) Monitor vital signs for any changes from initial findings throughout care.(3) Reassess and record vital signs at least every 15 minutes in a stable patient and at least every 5 minutes in an unstable patient.(4) Reassess and record vital signs after all medical interventions. 1-6. SAMPLE HISTORY SAMPLE is an acronym used to help determine a patient's history of the current illness. SAMPLE history is very important in that will help you to determine some of the patient's key complaints. In the medical patient, a good history will help determine about 80 percent of the indications of what illness you are dealing with. During the SAMPLE history, it is also important to determine what allergies and medications the patient may have. This is a very important step in the treatment of any patient. a. Signs and Symptoms. Signs are the things you can see about the patient's condition. Symptoms are what the patient tells you about his condition. Use OPQRST to help determine the patient's history. (1) O -- Onset of the current condition, What were you doing when this happened? Did it come on suddenly? Did it come on slowly? (2) P-- Provokes. What makes this condition better or worse? Did this get better when you rested? Took a medication? (3) Q -- Quality. What is the quality of the pain? Have the patient describe in his own words what the pain feels like (stabbing, pressure, tearing, crushing, etc.). Try not to lead the patient by asking questions like, "Is it a stabbing pain?" (4) R -- Radiation. Does the pain radiate? Or is it located in one specific area? (5) S -- Severity. This is usually assessed by having the patient rate the pain on a scale of 1 to 10 with 10 being the worse. It is necessary to ask the patient about the worst pain they have ever felt to obtain a good basis for their pain threshold and previous pain exposure. (6) T -- Time, How long has it been since the pain started? b. Allergies. Is the patient allergic to medications, food, or other substances? c. Medication. What medications is the patient currently taking? Make sure to ask about over-the-counter medications, herbal medications, and supplements that the patient may be taking. d. Pertinent Past History. Does the patient have any pertinent medical history? Anything that the patient may feel is applicable to the current illness or injury? e. Last Oral Intake. When did the patient last eat or drink? f. Events Leading to the Injury or Illness. What events lead to this incident? What where you doing just before the event happened or started? INTRODUCTION Improved understanding of the pathophysiology and the advances made in burn management over the past 50 years have contributed immensely to the dramatic rise in the survival and reduced morbidity from major burn injuries. The aim has always been to achieve wound healing as early as possible and to minimise the morbidity. The efforts towards this should commence right from the scene of the burn accident, at the time of first interaction between the casualty and the first responder. The care the victim receives within the first few hours after sustaining a burn injury largely determines the final outcome of the management. A burn accident can happen at any place and at any time and medical personnel are usually not around. So, all people should be aware of what the first aid for burns is, and should be able to administer the same, immediately, at the site of the accident. This is most often provided by non-professionals, i.e., friends, relatives, bystanders, etc., at the accident site. In fire accidents, by the time the expert medical help arrives, the injury might become lifethreatening. There is no time to wait for seeking an expert help. The first aid has to be provided by the bystanders, who become the “first responders”. Every second is precious and the quicker the first aid is provided, the minimal is the extent of damage. For any fire to happen, three essential ingredients required are oxygen, fuel and ignition. Without these, the burning process cannot begin (or continue). Eliminating any of the three components necessary for combustion will extinguish/prevent the fire.[1] In the case of human body, the body tissues provide the fuel, oxygen is present in plenty in the atmosphere and the ignition is provided by the spark from any source. Pre-hospital care or on-site management” implies the management at the site of the trauma and constitutes the major part of the first aid. It begins at the scene of the accident and concludes when specialised/institutional medical care is obtained. It must be readily available, easy to use by the general public, halt the progression of the injury and not hinder professional examination or treatment of the wound at a later date.[2] Promptly administered pre-hospital care, in an effective and systematic manner reduces the extent and limits the depth of burns, thus minimising the morbidity. Here, two important points must be borne in mind:
Thermal burns
Arrange transport to the medical facility
Guidelines for first aid for chemical burns:
First aid for chemical burns of the eye/cornea
Electrical burns Electrical burns occur due to contact with live electric wires or lightning. These injuries are classified under major burns and the victim needs to be transported to a burns care facility. Tetanic contractions induced in the muscle prevent the victim to free himself/ herself from the source. Strong contractions of flexor muscle group in upper limb tend to maintain the contact and it may cause muscle injury, joint dislocations and fractures.[18]
First aid in smoke inhalation primarily comprises:
Griffith (1985) identified the following five groups for victims of burn disasters:[27]
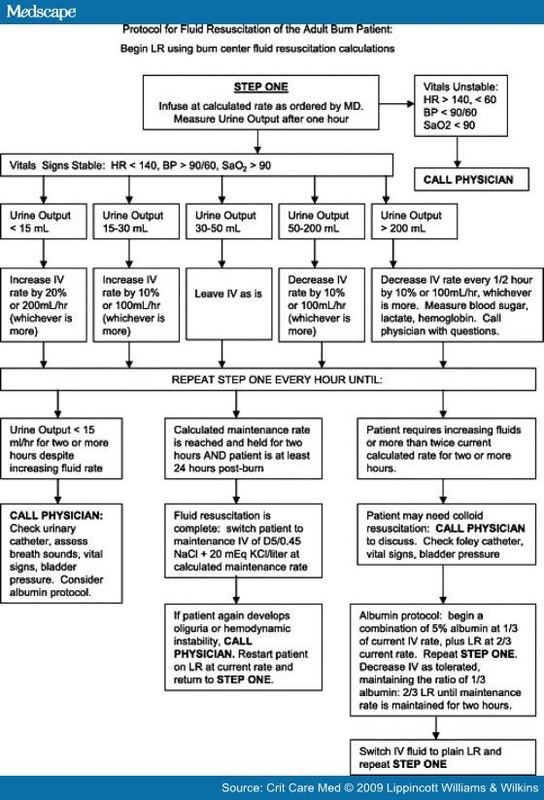
AbstractObjective: The goal of this concise review is to provide an overview of some of the most important resuscitation and monitoring issues and approaches that are unique to burn patients compared with the general intensive care unit population. Study Selection: Consensus conference findings, clinical trials, and expert medical opinion regarding care of the critically burned patient were gathered and reviewed. Studies focusing on burn shock, resuscitation goals, monitoring tools, and current recommendations for initial burn care were examined. Conclusions: The critically burned patient differs from other critically ill patients in many ways, the most important being the necessity of a team approach to patient care. The burn patient is best cared for in a dedicated burn center where resuscitation and monitoring concentrate on the pathophysiology of burns, inhalation injury, and edema formation. Early operative intervention and wound closure, metabolic interventions, early enteral nutrition, and intensive glucose control have led to continued improvements in outcome. Prevention of complications such as hypothermia and compartment syndromes is part of burn critical care. The myriad areas where standards and guidelines are currently determined only by expert opinion will become driven by level 1 data only by continued research into the critical care of the burn patient. IntroductionMajor strides in understanding the principles of burn care over the last half century have resulted in improved survival rates, shorter hospital stays, and decreases in morbidity and mortality rates due to the development of resuscitation protocols, improved respiratory support, support of the hypermetabolic response, infection control, early burn wound closure, and early enteral nutrition.[1] Critical care of the burn patient requires the participation of every discipline in the hospital. Resuscitation GoalsEffective fluid resuscitation is one of the cornerstones of modern burn care and perhaps the advance that has most directly improved patient survival. Proper fluid resuscitation aims to anticipate and prevent rather than to treat burn shock.[2–4] Resuscitation of burn shock cannot hope to achieve complete normalization of physiologic variables because the burn injury leads to ongoing cellular and hormonal responses. The obvious challenge is to provide enough fluid replacement to maintain perfusion without causing fluid overload.[3, 5–17] Without effective and rapid intervention, hypovolemia/shock will develop if the burns involve > 15% to 20% total body surface area (TBSA).[18] Delay in fluid resuscitation beyond 2 hrs of the burn injury complicates resuscitation and increases mortality.[7, 16] The consequences of excessive resuscitation and fluid overload are as deleterious as those of under-resuscitation: pulmonary edema, myocardial edema, conversion of superficial into deep burns, the need for fasciotomies in unburned limbs, and abdominal compartment syndrome.[5, 19–22] A Lund-Browder chart should be completed at the time of admission to calculate the TBSA burn.[1] Burn Shock PathophysiologyBurn shock is a unique combination of distributive and hypovolemic shock[5, 22–26] manifested by intravascular volume depletion, low pulmonary artery occlusion pressures, elevated systemic vascular resistance, and depressed cardiac output.[23, 27] Reduced cardiac output is a combined result of decreased plasma volume, increased afterload, and decreased contractility.[4] Studies suggest that impaired myocardial contractility is likely caused by circulating mediators such as tumor necrosis factor-α,[28, 29] however impaired Ca+2 at the cellular level is most likely involved as well.[30] The exact mechanisms of altered cardiac mechanical function remain unclear and are most likely multifactorial.[5, 30, 31] Virtually all components that control fluid and protein loss from the vascular space are altered after a burn.[25] Immediately after burn injury, the systemic microcirculation loses its vessel wall integrity and proteins are lost into the interstitium.[5, 17, 32] This protein loss causes the intravascular colloid osmotic pressure to drop precipitously and allows fluid to escape from the circulatory system.[5, 32] There is a marked transient decrease in interstitial pressure caused by the release of osmotically active particles, causing a vacuum effect that sucks in fluid from the plasma space. There is a marked increase in fluid flux into the interstitium caused by a combination of the sudden decrease in interstitial pressure, an increase in capillary permeability to protein, and a further imbalance in hydrostatic and oncotic forces favoring the fluid movement into the interstitium.[25] The outcome is a dramatic outpouring of fluids, electrolytes, and proteins into the interstitium with rapid equilibrium of intravascular and interstitial compartments.[17] These changes are reflected in loss of circulating plasma volume, hemoconcentration, massive edema formation, decreased urine output, and depressed cardiovascular function.[23] What actually changes is the volume of each fluid compartment, with intracellular and interstitial volumes increasing at the expense of plasma and blood volume.[17] Functional plasma volume in burn tissue can be restored only with expansion of the extracellular space.[33] Most edema occurs locally at the burn site and is maximal at 24 hrs postinjury.[5, 14, 17, 18, 25, 33, 34] The rate and extent of edema formation in major burn injury far exceed the intended beneficial effect of inflammatory system activation.[21, 25] The edema itself results in tissue hypoxia and increased tissue pressure with circumferential injuries. Aggressive fluid therapy can correct the hypovolemia but will accentuate the edema process.[21, 25, 35, 36] Resuscitation FormulasAdequate resuscitation from burn shock is the single most important therapeutic intervention in burn treatment. Due to a paucity of evidence-based literature, burn resuscitation remains an area of clinical practice driven primarily by local custom of treating burn units.[20] The only issue exempt from debate is that fluid administration is universally advocated.[22, 37] Each patient will react uniquely to burn injury depending on age, depth of burn, concurrent inhalation injury, preexisting comorbidities, and associated injuries. Formulas should be regarded as a resuscitation guideline; fluid administration has to be adjusted to individual patient needs. Of the numerous formulas for fluid resuscitation, none is optimal regarding volume, composition, or infusion rate.[2, 4–6, 12, 15, 17, 32] Lactated Ringer's solution most closely resembles normal body fluids. Factors that influence fluid requirements during resuscitation besides TBSA burn include burn depth, inhalation injury, associated injuries, age, delay in resuscitation, need for escharotomies/fasciotomies, and use of alcohol or drugs.[34] The Parkland formula has been renamed the Consensus formula because it is the most widely used resuscitation guideline. The Advanced Burn Life Support curriculum supports the use of the Consensus formula for resuscitation in burn injury.[32] Simply put, it is 4 mL/kg per percentage TBSA, describing the amount of lactated Ringer's solution required in the first 24 hrs after burn injury, where kg represents patient weight, and percentage TBSA is the size of the burn injury. Starting from the time of burn injury, half of the fluid is given in the first 8 hrs and the remaining half is given over the next 16 hrs. The rapid determination of percentage TBSA burn and calculation of the fluid requirements can be difficult and often incorrect when the person treating these burns is an inexperienced clinician. The substantial errors in estimating burn extent and depth result in significant under- or overcalculation of fluid requirements.[17, 18, 38, 39] Most doctors outside burn centers have infrequent experience with major burn management and a relative lack of sufficient knowledge regarding such management.[3, 17, 36, 38] Even among burn center physicians, there is considerable variability in determining the amount of fluids to be administered during the resuscitation period. There has not been a clinical advantage with colloids.[5, 12, 40] One study showed a decreased risk of death when albumin was used during resuscitation,[20] but the difference did not achieve statistical significance. A meta-analysis comparing albumin to crystalloid showed a 2.4-fold increased risk of death with albumin.[24] Hypertonic saline has also had disappointing results, with a four-fold increase in renal failure and twice the mortality of patients given lactated Ringer's solution.[41] Hypertonic saline does not routinely have a place in burn resuscitation.[22] Fresh frozen plasma should not be used as a volume expander, according to new policies on blood product delivery.[24] Due to the risk of blood-borne infectious transmission,[5] the American Burn Association Practice Guidelines for Burn Shock Resuscitation do not recommend the use of fresh frozen plasma without active bleeding or coagulopathy outside of a clinical trial, when other choices are available.[4] Depletion of limited blood bank reserves is another deterrent to using fresh frozen plasma in burn resuscitation.[5] output-related variable to manipulate in burn patients is preload. Pulmonary artery occlusion pressure and central venous pressure are not good indicators of preload.[5] As long as other signs of adequate tissue perfusion are normal, the temptation to normalize filling pressures should be avoided.[32] The use of end points demonstrating the adequacy of oxygen delivery has not yet found a place in the management of burn shock.[11, 23, 49] Laboratory Studies. Although the initial lactate is a strong predictor of mortality,[5, 20, 50] it is not clear how serum lactate can be used as a resuscitation end point.[32, 50, 51] Although lactate and base deficit (BD) are resuscitation markers that act as independent variables,[50–52] there is a low correlation between urinary output, mean arterial pressure, serum lactate, and base deficit.[51] Serum lactate trends provide greater information regarding the homeostatic status.[53, 54] Determinations of BD do not demonstrate the same predictive power; the effect of specific correction of the BD during fluid resuscitation is unknown.[13, 50, 52] There are insufficient data to make recommendations on the use of BD or lactate as resuscitation guidelines during burn resuscitation or as independent predictors of outcome in patients with large burns.[5, 32, 51, 55] Hematocrits of 55% to 60% are not uncommon in the early postburn period and cannot be used to monitor fluid resuscitation.
Resuscitation End Points. End points of resuscitation have been the subject of numerous strategies with conflicting results.[5, 13, 15, 16, 19, 22–24] Many authors feel that urine output[34] and traditional vital signs (heart rate and mean arterial pressure) are too insensitive to ensure appropriate fluid replacement in burn injuries.[11, 32, 49, 51] In children, trends in heart rate, blood pressure, and capillary refill toward normal are more reasonable therapeutic end points.[19] In adults, arterial blood pressure is relatively insensitive to the adequacy of fluid replacement; pulse rate is more helpful. In older patients, pulse rate becomes less reliable. Urine output can be taken to reflect organ perfusion; however, urine must be nonglycosuric to be accurate.[36] Hypertonic saline can increase urine output due to an osmotic diuresis that does not accurately reflect volume status.[33] Although urine output does not precisely mirror renal blood flow, it remains the most readily accessible and easily monitored index of resuscitation.[35, 56] Fluid Creep. The use of excessive volumes for resuscitation is being documented with increasing frequency in many burn centers.[39, 57] Burn care providers have become more aggressive with the administration of benzodiazepines and narcotics, which may result in additional fluid demands.[18, 20, 56, 58–60] Outreach education in burn care has contributed to a now-common problem of excessive resuscitation given by first responders and non-burn physicians. Thus, many patients arrive at a burn center having received most of their first 8-hr Consensus formula requirements in just an hour or two.[39] Vitamin C Resuscitation. The landmark study by Tanaka et al showed that high dose ascorbic acid during the initial 24 hrs post burn reduced fluid requirements by 40%, reduced burn tissue water content 50%, and reduced ventilator days.[61, 62] The clinical benefits led to a clear reduction in edema and body weight gain and were associated with reduced respiratory impairment and reduced requirement for mechanical ventilation.[36, 61, 62] Although not in mainstream use, the findings are meaningful to experienced burn care practitioners. Inhalation Injury. The combination of a body burn and smoke inhalation produces a marked increase in mortality and morbidity.[63, 64] Burn patients with inhalation injury have been shown to require increased fluids during resuscitation.[1, 8, 15, 37, 65] Navar et al[66] found that the presence of inhalation injury was associated with a 44% increase in fluid requirements, which was remarkably uniform across all age groups and burn sizes. The degree of lung dysfunction caused by a smoke inhalation injury is accentuated by the presence of even a small body burn.[25, 36, 64, 65] Acute upper airway obstruction occurs in 20% to 33% of hospitalized burn patients with inhalation injury and is a major hazard because of the possibility of rapid progression from mild pharyngeal edema to complete upper airway obstruction.[67] Patients presenting with stridor should be intubated on presentation. Patients at risk of requiring early intubation include those with a history of being in an enclosed space with or without facial burns, history of unconsciousness, carbonaceous sputum, voice change, or complaints of a "lump in the throat." In isolation, these factors do not predict the need for intubation, but the more signs present, the more elevated the risk. A carboxyhemoglobin level taken within 1 hr after injury is strongly indicative of smoke inhalation if > 10%.[3] If there is a significant cutaneous burn requiring resuscitation, the need for intubation will be greater. The small cross-sectional diameter of the pediatric airway places children at higher risk of requiring emergent intubation. If intubation is needed, the most experienced clinician in airway management should perform endotracheal intubation.[67] Intubation itself is not without risk so should not be undertaken routinely simply because there are facial burns. The care of inhalation injury remains supportive. Even the gold standard of bronchoscopy within the first 24 hrs of admission cannot accurately predict the severity of inhalation injury. For patients with inhalation injury, no ideal ventilator strategy has emerged.[67] According to the American College of Chest Physicians, recommendations for mechanical ventilation serve as general guidelines: Use a ventilator mode that is capable of supporting oxygenation and ventilation that the clinician has experience using, limit plateau pressures to < 35 cm H2O, allow Pco2 to increase if needed to minimize plateau pressures, and use the appropriate level of positive end-expiratory pressure.[68] Roughly 70% of patients with inhalation injury will develop ventilator-associated pneumonia. Routine pneumonia prevention strategies should include elevating the head of the bed 30°, turning the patient side to side every 2 hrs, oral care every 6 hrs, and gastrointestinal prophylaxis. Prophylactic antibiotics have no role and actually increase infection rates. For patients who fail to respond to maximal conventional therapy, consider extracorporeal membrane oxygenation as a rescue therapy for patients with acute respiratory failure who are expected to die otherwise.[69] Preventable ComplicationsHypothermia. The profoundly adverse effects of hypothermia cannot be overstated. Strategies to vigorously prevent hypothermia include a warmed room, warmed inspired air, warming blankets, and countercurrent heat exchangers for infused fluids. Metabolic responses can be minimized by treating the patient in a thermoneutral environment (32°C).[3] During hydrotherapy, in the operating room, and in the burn unit, keep the room temperature at ≥85°F to minimize heat loss and decrease metabolic rate. Compartment Syndromes. A life-threatening complication caused by high-volume resuscitation is abdominal compartment syndrome (ACS),[24] defined as intra-abdominal pressure ≥20 mm Hg plus at least one new organ dysfunction.[70] ACS has been associated with renal impairment, gut ischemia, and cardiac and pulmonary malperfusion. Clinical manifestations include tense abdomen, decreased pulmonary compliance, hypercapnia, and oliguria. Simply monitoring urine output is insufficiently sensitive or specific to diagnose ACS.[36, 71, 72] Vigilant monitoring and aggressive treatment should be instituted to avoid this deadly complication.[71, 72] Appropriate intravascular volume, appropriate body positioning, pain management, sedation, nasogastric decompression if appropriate, chemical paralysis if required, and torso escharotomy are all interventions to increase abdominal wall compliance and decrease intra-abdominal pressures.[72, 73] Bladder pressure monitoring should be initiated as part of the burn fluid resuscitation protocol in every patient with > 30% TBSA burn.[5, 24, 73] Patients who receive > 250 mL/kg of crystalloid in the first 24 hrs will likely require abdominal decompression.[15] Percutaneous abdominal decompression is a minimally invasive procedure that should be performed before resorting to laparotomy.[71, 74] The International Conference of Experts on Intra-abdominal Hypertension and Abdominal Compartment Syndrome recommends that if less invasive maneuvers fail, decompressive laparotomy should be performed in patients with ACS that is refractory to other treatment options.[72] The reported mortality rates for decompressive laparotomy for ACS can be as high as 88%[71] to 100%.[74] Extremity compartment syndromes can also result from extensive edema formation. Patients may require escharotomies, fasciotomies, or both for the release of extremity compartment syndrome.[36, 75] Patients with circumferential full-thickness burns are also at risk of requiring escharotomies.[35] Impaired capillary refill, paresthesia in the involved extremity, and increased pain develop earlier than decreased pulses. The orbit is a compartment limited to expansion and may require lateral canthotomy to successfully reduce intraocular pressure to normal.[76] Deep Venous Thrombosis. The incidence of deep venous thrombosis in burn patients is estimated to be 1% to 23%.[77] In the absence of level 1 evidence, deep venous thrombosis chemoprophylaxis is routinely practiced in many burn centers. Heparin-induced Thrombocytopenia. Early thrombocytopenia occurs in the postburn course in patients with extensive injury. Problems after burn injury such as pulmonary infections, multiorgan failure, sepsis, and bleeding disorders accentuate this trend. As in nonburn patients, careful observance for thrombocytopenia after the first week of hospitalization will alert the practitioner to make the diagnosis in burn patients.[78, 79] Although the incidence of heparin-induced thrombocytopenia was relatively low (1.6%) in one study,[79] the complications in those patients were profound, including arterial and deep venous thromboses and increased number of surgical procedures.[79] Neutropenia. Transient leukopenia is common, primarily due to a decreased neutrophil count. Maximal white blood cell depression occurs several days after admission with rebound to normal a few days later. Use of silver sulfadiazine has been associated with this transient leukopenia; resolution is independent of continued silver sulfadiazine.[1] Stress Ulcers. Level 1 data exists that patients with major burn injuries are at risk for stress ulcers and should receive routine prophylaxis beginning at admission.[80] Adrenal Insufficiency. Although absolute adrenal insufficiency occurs in up to 36% of patients with major burns, there is no correlation between response to corticotropin stimulation and survival. Those with massive burns have higher cortisol levels but may be resistant to serum cortisol increases in response to stimulation. The clinical relevance of this finding has not been established.[81, 82] Infection/Inflammation/SepsisConsensus Paper on Sepsis and Infection-related Diagnoses. Current definitions for sepsis and infection have many criteria routinely found in patients with extensive burns without infection/sepsis (e.g., fever, tachycardia, tachypnea, leukocytosis). Burn experts recently developed standardized definitions for sepsis and infection-related diagnoses in burn patients from which I will summarize key discussion points and recommendations.[78] Patients with large burns have a baseline temperature reset to 38.5°C, and tachycardia and tachypnea may persist for months. Continuous exposure to inflammatory mediators leads to significant changes in the white blood cell count, making leukocytosis a poor indicator of sepsis. Use other clues as signs of infection or sepsis such as increased fluid requirements, decreasing platelet counts > 3 days after burn injury, altered mental status, worsening pulmonary status, and impaired renal function. The term systemic inflammatory response syndrome should not be applied to burn patients because patients with large burns are in a state of chronic systemic inflammatory stimulation.[78] Any infection in a burn patient should be considered to be from the central venous catheter until proven otherwise.[78] Central catheters should be changed to a new site every 3 days to minimize bloodstream infections.[83] Although prophylactic systemic antibiotics have no role in thermal injury, topical antimicrobial therapy is efficacious.[1] Systemic antibiotic therapy should be culture directed and administered for the shortest time possible. Metabolism/NutritionEnteral Nutrition. As hypermetabolism can lead to doubling of the normal resting energy expenditure, enteral nutrition should be started as soon as resuscitation is underway with a transpyloric feeding tube. Patients with burns > 20% TBSA will be unable to meet their nutritional needs with oral intake alone. Patients fed early have significantly enhanced wound healing and shorter hospital stays.[84] In the rare case that precludes use of the gastrointestinal tract, parenteral nutrition should be used only until the gastrointestinal tract is functioning. Endocrine and Glucose Monitoring. Strict glucose control of 80–110 mg/dL can be achieved using an intensive insulin therapy protocol, leading to decreased infectious complications and mortality rates.[85, 86] Anabolic Steroids. Severe burn injuries induce a hypermetabolic response, which leads to catabolism. Anabolic androgenic steroids such as oxandrolone promote protein synthesis, nitrogen retention, skeletal muscle growth, and decreased wound healing time. Burn patients receiving oxandrolone regain weight and lean mass two to three times faster than with nutrition alone.[87] β-Blockade. β-blockers after severe burns decrease heart rate, resulting in reduced cardiac index and decreased supraphysiologic thermogenesis.[3, 88] In children with burns, treatment with propranolol during hospitalization attenuates hypermetabolism and reverses muscle-protein catabolism. Propranolol is given to achieve a 20% decrease in heart rate of each patient compared with the 24-hr average heart rate immediately before administration.[88] Additional TherapiesWound Management. The primary goal for burn wound management is to close the wound as soon as possible, beginning at the time of injury. Burn centers are uniquely set up to provide optimal wound care. Beginning on admission and then daily, hydrotherapy is routine, involving washing the entire patient with chlorhexidine and warm tap water. The goal is to gently debride the nonviable tissue while leaving any newly formed dermis/epidermis. The practice of immersion in large tanks or other standing bodies of water has fallen out of favor, as bacteria from the fecal fallout zone quickly colonize the entire burn wound. Once the wound is clean, topical antimicrobial agents limit bacterial proliferation and fungal colonization in the burn wound.[26] Silver sulfadiazine is the most commonly used topical antimicrobial, being readily available, affordable, and well tolerated by the patient. There are also silver-containing sheets and compounds that may be placed on partial thickness burns and remain in place for up to 7 days. For patients with full-thickness burns, prompt surgical excision of the eschar and allografting in patients with large burns, or autografting in patients with smaller burns, contributes to reduced morbidity and mortality.[26] A host of temporary wound coverage products are available. Pain Management. Burn patients may experience pain that is multifaceted and constantly changing as the individual undergoes repeated procedures and wound manipulation. Inconsistent and inadequate pain management has been well documented. Although there is no universal treatment standard for pain management, opioid doses often significantly exceed recommended standard dosing guidelines.[60, 89] Practice Management Guidelines for the Management of Pain by the Committee on the Organization and Delivery of Burn Care of the American Burn Association recommends that once intravenous access is obtained and resuscitation started, intravenous opioids should be administered. Background pain is best managed through the use of long-acting analgesic agents. Breakthrough pain is addressed with short-acting agents via an appropriate route.[89] Ketamine can be used for extensive burn dressing changes and procedures such as escharotomies. Anxiolytics such as benzodiazepines decrease background and procedural pain.[89] For patients requiring mechanical ventilation, a propofol infusion will provide sedation but not analgesia. All medications should be given intravenously, orally, or rectally due to erratic absorption with intramuscular/subcutaneous administration. Physiotherapy. Rehabilitation therapy begins at admission to maximize functional recovery. Burn patients require special positioning and splinting, early mobilization, strengthening and endurance exercises to promote healing.[1] Transfer Criteria. The American Burn Association has established criteria for burn patients who should be acutely transferred to a burn center: > 10% TBSA partial thickness burns, any size full-thickness burn, burns to special areas of function or cosmesis, inhalation injury, serious chemical injury, electrical injury including lightning, burns with trauma where burns are the major problem, pediatric burns if the referring hospital has no special pediatric capabilities, and smaller burns in patients with multiple comorbidities.[ ConclusionsNot many topics in acute burn care are more hotly debated than fluid resuscitation and monitoring. Burn management is still not evidence based as in many areas of acute medicine.[24] However, there does seem to be agreement among burns surgeons that: 1) the Consensus formula provides for a hypovolemic resuscitation; 2) patients with inhalation injury will require more fluid than that prescribed by the Consensus formula; and 3) over-resuscitation leads to excessive burn edema, abdominal compartment syndrome, need for fasciotomies on unburned limbs, pulmonary edema, and prolongation of mechanical ventilation. Type of monitoring to use during the early resuscitation period remains controversial in part because current end points have not yet been demonstrated to reflect tissue perfusion status independently and accurately.[5, 91] Vital signs and urine output in burn patients do not fulfill these criteria.[14] Defining better end points of resuscitation to avoid excessive volume administration is a high priority for future investigations.[4] Future improvements in managing burn shock will include a complex ballet that includes pharmacologic interventions, rapid surgical removal of necrotic tissue, and a dynamic range of fluid types and rates of delivery. The continuing challenge for burn clinicians and researchers is to collaborate in large multicenter studies to critically evaluate and establish resuscitation end points and therapies.[5, 36] Blood pressure may be the vital sign we measure the most and understand the least1. You’re using the wrong-sized cuff
The most common error when using indirect blood pressure measuring equipment is using an incorrectly sized cuff. A BP cuff that is too large will give falsely low readings, while an overly small cuff will provide readings that are falsely high. The American Heart Association (AHA) publishes guidelines for blood pressure measurement,[2] recommending that the bladder length and width (the inflatable portion of the cuff) should be 80 percent and 40 percent respectively, of arm circumference. Most practitioners find measuring bladder and arm circumference to be overly time consuming, so they don’t do it. The most practical way to quickly and properly size a BP cuff is to pick a cuff that covers two-thirds of the distance between your patient’s elbow and shoulder. Carrying at least three cuff sizes (large adult, regular adult, and pediatric) will fit the majority of the adult population. Multiple smaller sizes are needed if you frequently treat pediatric patients. Korotkoff sounds are the noises heard through a stethoscope during cuff deflation. They occur in 5 phases:
2. You’ve incorrectly positioned your patient’s body The second most common error in BP measurement is incorrect limb position. To accurately assess blood flow in an extremity, influences of gravity must be eliminated. The standard reference level for measurement of blood pressure by any technique (direct or indirect) is at the level of the heart. When using a cuff, the arm (or leg) where the cuff is applied must be at mid-heart level. Measuring BP in an extremity positioned above heart level will provide a falsely low BP whereas falsely high readings will be obtained whenever a limb is positioned below heart level. Errors can be significant — typically 2 mmHg for each inch the extremity is above or below heart level. A seated upright position provides the most accurate blood pressure, as long as the arm in which the pressure is taken remains at the patient’s side. Patients lying on their side, or in other positions, can pose problems for accurate pressure measurement. To correctly assess BP in a side lying patient, hold the BP cuff extremity at mid heart level while taking the pressure. In seated patients, be certain to leave the arm at the patient’s side. Arterial pressure transducers are subject to similar inaccuracies when the transducer is not positioned at mid-heart level. This location, referred to as the phlebostatic axis, is located at the intersection of the fourth intercostal space and mid-chest level (halfway between the anterior and posterior chest surfaces. Note that the mid-axillary line is often not at mid-chest level in patients with kyphosis or COPD, and therefore should not be used as a landmark. Incorrect leveling is the primary source of error in direct pressure measurement with each inch the transducer is misleveled causing a 1.86 mmHg measurement error. When above the phlebostatic axis, reported values will be lower than actual; when below the phlebostatic axis, reported values will be higher than actual. 3. You’ve placed the cuff incorrectly The standard for blood pressure cuff placement is the upper arm using a cuff on bare skin with a stethoscope placed at the elbow fold over the brachial artery. The patient should be sitting, with the arm supported at mid heart level, legs uncrossed, and not talking. Measurements can be made at other locations such as the wrist, fingers, feet, and calves but will produce varied readings depending on distance from the heart. The mean pressure, interestingly, varies little between the aorta and peripheral arteries, while the systolic pressure increases and the diastolic decreases in the more distal vessels. Crossing the legs increases systolic blood pressure by 2 to 8 mmHg. About 20 percent of the population has differences of more than 10 mmHg pressure between the right and left arms. In cases where significant differences are observed, treatment decisions should be based on the higher of the two pressures. 4. Your readings exhibit ‘prejudice’ Prejudice for normal readings significantly contributes to inaccuracies in blood pressure measurement. No doubt, you’d be suspicious if a fellow EMT reported blood pressures of 120/80 on three patients in a row. As creatures of habit, human beings expect to hear sounds at certain times and when extraneous interference makes a blood pressure difficult to obtain, there is considerable tendency to “hear” a normal blood pressure. Orthostatic hypotension is defined as a decrease in systolic blood pressure of 20 mmHg or more, or diastolic blood pressure decrease of 10 mmHg or more measured after three minutes of standing quietly. There are circumstances when BP measurement is simply not possible. For many years, trauma resuscitation guidelines taught that rough estimates of systolic BP (SBP) could be made by assessing pulses. Presence of a radial pulse was thought to correlate with an SBP of at least 80 mmHg, a femoral pulse with an SBP of at least 70, and a palpable carotid pulse with an SBP over 60. In recent years, vascular surgery and trauma studies have shown this method to be poorly predictive of actual blood pressure[3]. Noise is a factor that can also interfere with BP measurement. Many ALS units carry doppler units that measure blood flow with ultrasound waves. Doppler units amplify sound and are useful in high noise environments. BP by palpation or obtaining the systolic value by palpating a distal pulse while deflating the blood pressure cuff generally comes within 10 – 20 mmHg of an auscultated reading. A pulse oximeter waveform can also be used to measure return of blood flow while deflating a BP cuff, and is as accurate as pressures obtained by palpation. In patients with circulatory assist devices that produce non-pulsatile flow such as left ventricular assist devices (LVADs), the only indirect means of measuring flow requires use of a doppler. The return of flow signals over the brachial artery during deflation of a blood pressure cuff in an LVAD patient signifies the mean arterial pressure (MAP). While a normal MAP in adults ranges from 70 to 105 mmHg, LVADs do not function optimally against higher afterload, so mean pressures of less than 90 are often desirable. Clothing, patient access, and cuff size are obstacles that frequently interfere with conventional BP measurement. Consider using alternate sites such as placing the BP cuff on your patient’s lower arm above the wrist while auscultating or palpating their radial artery. This is particularly useful in bariatric patients when an appropriately sized cuff is not available for the upper arm. The thigh or lower leg can be used in a similar fashion (in conjunction with a pulse point distal to the cuff). All of these locations are routinely used to monitor BP in hospital settings and generally provide results only slightly different from traditional measurements in the upper arm. 5. You’re not factoring in electronic units correctly Electronic blood pressure units also called Non Invasive Blood Pressure (NIBP) machines, sense air pressure changes in the cuff caused by blood flowing through the BP cuff extremity. Sensors estimate the Mean Arterial Pressure (MAP) and the patient’s pulse rate. Software in the machine uses these two values to calculate the systolic and diastolic BP. To assure accuracy from electronic units, it is important to verify the displayed pulse with an actual patient pulse. Differences of more than 10 percent will seriously alter the unit’s calculations and produce incorrect systolic and diastolic values on the display screen. Given that MAP is the only pressure actually measured by an NIBP, and since MAP varies little throughout the body, it makes sense to use this number for treatment decisions. A normal adult MAP ranges from 70 to 105 mmHg. As the organ most sensitive to pressure, the kidneys typically require an MAP above 60 to stay alive, and sustain irreversible damage beyond 20 minutes below that in most adults. Because individual requirements vary, most clinicians consider a MAP of 70 as a reasonable lower limit for their adult patients. Increased use of NIBP devices, coupled with recognition that their displayed systolic and diastolic values are calculated while only the mean is actually measured, have led clinicians to pay much more attention to MAPs than in the past. Many progressive hospitals order sets and prehospital BLS and ALS protocols have begun to treat MAPs rather than systolic blood pressures. Finally, and especially in the critical care transport environment, providers will encounter patients with significant variations between NIBP (indirect) and arterial line (direct) measured blood pressure values. In the past, depending on patient condition, providers have elected to use one measuring device over another, often without clear rationale besides a belief that the selected device was providing more accurate blood pressure information. In 2013, a group of ICU researchers published an analysis of 27,022 simultaneous art line and NIBP measurements obtained in 852 patients[4]. When comparing the a-line and NIBP readings, the researchers were able to determine that, in hypotensive states, the NIBP significant overestimated the systolic blood pressure when compared to the arterial line, and this difference increased as patients became more hypotensive. At the same time, the mean arterial pressures (MAPs) consistently correlated between the a-line and NIBP devices, regardless of pressure. The authors suggested that MAP is the most accurate value to trend and treat, regardless of whether BP is being measured with an arterial line or an NIBP. Additionally, supporting previously believed parameters for acute kidney injury (AKI) and mortality, the authors noted that a MAP below 60 mmHg was consistently associated with both AKI and increased mortality. Since 1930, blood pressure measurement has been a widely accepted tool for cardiovascular assessment. Even under the often adverse conditions encountered in the prehospital or transport environment, providers can accurately measure blood pressure if they understand the principles of blood flow and common sources that introduce error into the measurement process. |
Archives
October 2014
Categories
All
|
 RSS Feed
RSS Feed
